Bill Colone, a medical executive who has built a career on commercializing doctors’ ideas, has launched a new venture.
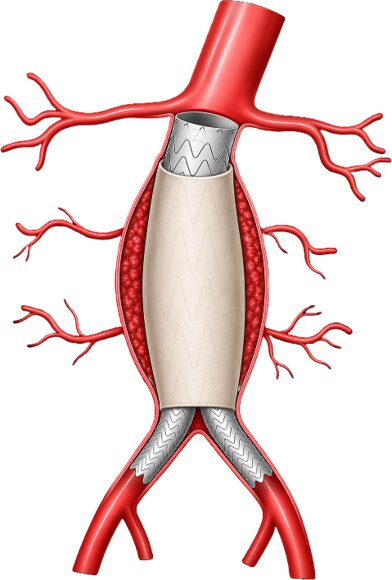
His new company Life Seal Vascular has built a device to solve an issue associated with aortic abdominal aneurysms, which 200,000 people are estimated to be diagnosed with in the U.S. every year.
The device aims to prevent “endoleaks”—a complication in which blood leaks into the aneurysm sac after a minimally invasive procedure.
“Over half of the reinterventions are due to this specific problem, and no one’s addressed it thus far,” Colone told the Business Journal. “So, we think this is a really large and growing market with very clear unmet need.”
It’s the latest project for Colone, who stepped down as CEO last April from Lake Forest-based Single Pass Inc., maker of “the first disposable biopsy cauterization device.” Colone was succeeded by John Zehren, who was already on the board of directors.
“I’m way more of an early-stage development guy, and we were just in full-on commercialization with Single Pass,” Colone said.
Promising Early Data
The aorta is the largest artery in the body and carries blood from the heart to the rest of the circulatory system. The lower part is the abdominal aorta.
An abdominal aortic aneurysm is similar to other aneurysms in the sense that the blood vessel wall weakens—due to reasons including high blood pressure, poor diet or cardiovascular disease—and dilates.
The minimally invasive procedure to treat the issue puts a new lining in the aorta to take pressure off the diseased tissue, which can lead to endoleaks.
“Those branch vessels that usually have blood flow going out now don’t have any pressure going out,” Colone said.
Life Seal Vascular’s solution, dubbed Cygnum, aims to prevent a specific type of endoleak where blood flows backward from the branch vessels into the aneurysm sac.
Before the lining is placed, a compressible material is delivered through a sheath to block backflow—adding just five minutes to the current procedure.
Seven patients, five in New Zealand and two in Japan, have been treated with Cygnum with none experiencing any endoleaks, according to Colone.
“The early patient data is looking very promising,” he said.
Single Pass Exit
One of the reasons Colone decided to pursue Life Seal Vascular was due to Single Pass’ quick growth, he said.
Single Pass, founded in 2021, received CE mark and FDA approval within five months of each other in 2024. Its device is now selling in approximately 20 countries, including parts of Asia, according to Colone.
“Sales are going great and I would expect that in a certain time period that somebody would try to acquire that company,” Colone said.
Colone said that he was first approached with the idea of a device to prevent endoleaks a few years ago by a vascular surgeon from the Netherlands named Dr. Michel Reijnen.
Colone asked four physicians he’s worked with in the past to be advisors to the company, including Jan Heyligers, Dai Yamanouchi, Chris Kwolek and Andrew Holden, who’s also chief medical officer. All four were also early investors in Life Seal Vascular.
“That gave me the kind of confirmation that I needed that this is really good technology,” Colone.
They have raised around $3.5 million, consisting mainly of clinicians and a venture capital group made up of vascular surgeons.
Colone said that the company plans to launch a $10 million Series A round to fund the initiation of a pivotal clinical trial to get investigational device exemption (IDE) approval.