
South Korea has built visible momentum in AI healthcare, with rising regulatory approvals and expanding policy support. Yet inside hospitals, adoption remains uneven. The issue is no longer whether AI works. It is whether hospitals can integrate it into daily practice, justify it financially, and scale it across systems. This gap is now shaping the next phase of Korea’s healthcare innovation strategy.
Korea’s AI Healthcare Momentum Is Real, but Still Incomplete
The healthcare AI ecosystem in South Korea has rapidly moved beyond early experimentation.
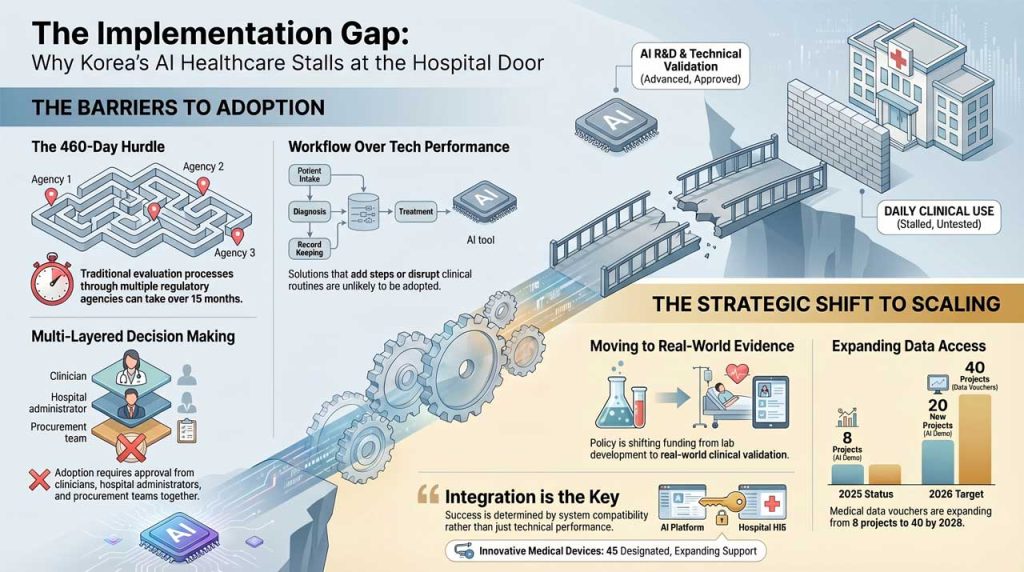
As of October 2025, 16 AI-based radiology technologies were already in clinical use, according to the Korean Journal of Radiology (2026). The Ministry of Food and Drug Safety also designated 45 innovative medical devices in 2025, including AI-based solutions. These figures confirm that Korea has established a functioning pipeline of AI medical technologies.
At the policy level, the Ministry of Health and Welfare has expanded support programs. In December 2025, it introduced initiatives aimed at addressing what it described as the “AI adoption gap in clinical settings.” This includes training programs and increased access to medical data for startups.
These moves signal a shift. Korea is no longer focused only on developing AI technologies. It is trying to make them actually usable inside real healthcare systems.
Where the System Breaks: From Validation to Real Use
Despite strong technical performance, many AI healthcare solutions fail to move beyond controlled environments.
Dr. Wonju Hwangbo, Founder & CEO of AORAEL and Director of Korea Society of Technology for Sustainable Future (KSTF), explained to KoreaTechDesk,
“The breakdown typically occurs not at the development stage, but at the point of integration into real clinical environments.”

Her observation aligns with recent policy direction. The government is now funding real-world data (RWD) and real-world evidence (RWE) generation, along with multicenter clinical validation programs. In 2026, it plans to launch 20 additional AI demonstration projects and expand medical data voucher support from 8 projects in 2025 to 40 in 2026.
This indicates that the main challenge lies between validation and deployment. AI systems may perform well in testing, but integrating them into hospital workflows introduces a different set of constraints.
Inside Hospitals: Adoption Is an Institutional Decision
On the other hand, it appears that hospital adoption is never determined by technology alone.
According to Dr. Hwangbo:
“Decisions are made through a multi-layered structure involving clinicians, hospital administrators, and procurement teams.”
Even when clinicians support a solution, adoption may still stall. Budget ownership, operational impact, and institutional priorities often override technical performance.
This is compounded by Korea’s multi-step evaluation system. A typical pathway involves regulatory approval by MFDS, clinical evaluation, and reimbursement assessment through agencies such as HIRA and NECA. The Korean Journal of Radiology notes that the traditional process can take around 460 days.
And so this layered structure creates significant friction. AI adoption becomes a negotiation across departments, rather than a straightforward deployment decision.
Workflow Friction: When AI Adds Complexity Instead of Value
Now, even after approval is granted, operational issues often limit sustained use.
Dr. Hwangbo highlights the core problem:
“If a solution increases workload or disrupts clinical routines, it is unlikely to be adopted.”
In practice, this can include:
- additional steps in diagnosis workflows
- unclear interpretation of AI outputs
- misalignment with existing hospital systems
And these are not technical failures. They are integration failures.
The government’s response reflects this. Training programs now focus on clinical usability and workforce readiness, suggesting that adoption depends as much on human systems as on software performance.
Reimbursement and Procurement Define Scale
At the same time, financial and institutional structures remain decisive.
HIRA introduced a temporary listing framework for innovative medical technologies, with the first listing in December 2023. According to its 2024 sustainability report, this expanded access to approximately 200,000 patients.
However, reimbursement systems are still evolving. Evaluation criteria, pricing structures, and post-listing monitoring remain under development.
The Ministry of Health and Welfare has responded with a post-approval commercialization support program in 2026. This includes:
- economic evaluation
- RWD and RWE accumulation
- hospital-company consortia for deployment
Dr. Hwangbo emphasizes the importance of these mechanisms:
“Without reimbursement, hospitals lack incentives to adopt new solutions.
Procurement systems also present barriers for startups.”
This places financial alignment at the center of adoption. Without it, even validated technologies struggle to scale.
Why Strong R&D Still Doesn’t Translate into Adoption
Korea’s challenge is not innovation output. It is system alignment.
Many AI healthcare projects remain at the pilot stage. According to Dr. Hwangbo:
“Projects focused only on technical performance tend to remain at the pilot stage. In healthcare, integration is the key to adoption.”
Government programs now reflect this reality. The expansion of data vouchers, demonstration projects, and post-approval support all target deployment rather than invention.
This suggests that Korea’s ecosystem still carries characteristics of an R&D-driven system. It produces strong technologies, but connecting them to institutional use remains complex.
How Korea Compares Globally
Korea’s position is not defined by technological capability alone.
Dr. Hwangbo notes,
“Korea is strong in R&D, infrastructure, and clinical expertise.
However, it is still developing integrated systems linking regulation, reimbursement, and market access.”
In markets such as the United States and parts of Europe, regulatory processes are increasingly linked with reimbursement pathways and stakeholder engagement earlier in the lifecycle.
Korea is moving in this direction. However, the need for new programs focused on post-approval deployment suggests that integration across systems is still incomplete.

What This Means for Startups and Investors
The implications reach well beyond Korea.
For founders, the lesson is structural. A strong product alone will not carry a company into real clinical use. What matters is how well the solution fits into daily medical practice, how clearly it connects to reimbursement, and how smoothly it works within existing hospital systems.
Meanwhile, investors face a similar shift in perspective. Technical performance is no longer the primary signal. The real questions sit deeper inside the system: how decisions are made within hospitals, who ultimately approves adoption, whether reimbursement can be secured, and how quickly a product can move past pilot testing into sustained use.
Dr. Hwangbo summarizes the core misunderstanding:
“The most common misunderstanding is that technical performance alone determines adoption.”
Because in practice, adoption depends on system compatibility.
The Real Challenge: Bridging Innovation and Deployment
South Korea’s AI healthcare sector is entering a new phase.
The country has demonstrated strong capabilities in developing and validating AI technologies. Regulatory frameworks are evolving, and policy support is expanding.
However, scaling these technologies requires alignment across:
- clinical workflows
- reimbursement systems
- procurement processes
- institutional decision-making
As Dr. Hwangbo notes:
“The challenge is not technological capability, but whether we can bridge the gap between innovation efficiency and deployment efficiency.”
This gap now defines the trajectory of Korea’s healthcare AI ecosystem.
Key Takeaway
- Korea has proven its ability to develop AI healthcare technologies, with 16 solutions already in clinical use.
- The main bottleneck is post-validation deployment, not early-stage innovation.
- Hospital adoption depends on workflow integration, institutional decision-making, and financial incentives.
- Reimbursement and procurement systems remain key constraints on scaling AI medical devices in Korea.
- Government policy is shifting toward real-world evidence, clinical deployment, and commercialization support.
- For global players, Korea illustrates a broader pattern:
AI healthcare success is determined not by how well the technology works, but by how well systems are aligned to use it.
🤝 Looking to connect with verified Korean companies building globally?
Explore curated company profiles and request direct introductions through beSUCCESS Connect.
– Stay Ahead in Korea’s Startup Scene –
Get real-time insights, funding updates, and policy shifts shaping Korea’s innovation ecosystem.
➡️ Follow KoreaTechDesk on LinkedIn, X (Twitter), Threads, Bluesky, Telegram, Facebook, and WhatsApp Channel.
Source link



